By country
By activity
- Rights (87)
- Emergency (587)
- Rehabilitation (392)
- Prevention (80)
- Inclusion (229)
- Health (156)
- Explosive weapons (310)
- Event (13)
- Covid-19 (5)
By publication date
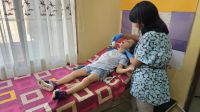
West Bank: Attending school in times of conflict is a challenge 04/17/24
Samar, 10, lives in a camp in the West Bank. She has multiple disabilities. Attending school is already a challenge, now exacerbated by the escalation of violence.
- Occupied Palestinian Territories
7 things you should know about the conflict in Sudan 04/15/24
On April 15 2023, an armed conflict broke out in Sudan. In May 2023, HI mobilized in Eastern Chad to support and care for Sudanese refugees fleeing the violence.
- Chad
Gaza: “We need double the amount of supplies” 04/08/24
Wala provides nursing care to people in their homes and shelters. She talks to us about her life, her work and the situation in Gaza.
- Occupied Palestinian Territories
Martine, a mine accident survivor, tells her story to raise awareness of the risks 04/05/24
Martine comes from Senegal. In 2009, she was injured in a mine explosion. Since then, she has been speaking out to raise awareness of the dangers of explosive devices and prevent further accidents.
- Senegal
Good news in March 2024 03/29/24
Good news in March at HI: rehabilitation services and orthopedic devices in Peru, Cambodia and the Central African Republic, and inclusion in schools in Kenya.
- International
Five years after Cyclone Idai, lessons learned from the disaster 03/11/24
In March 2019, Cyclone Idai struck Mozambique, leaving a trail of desolation in its wake. Five years on, lessons have been learned about the inclusiveness of aid and disaster prevention.
- Mozambique
Rehabilitation needs growing rapidly in Gaza 02/26/24
As violence rages in Gaza, the number of injured people is soaring. Many are at risk of permanent disability.
- Occupied Palestinian Territories
Good news of February 2024 02/26/24
Good news in February at HI: demining in Senegal, inclusive development in Venezuela, post-emergency rehabilitation in Morocco and advocacy in the Philippines.
- Philippines
- Morocco
- Senegal
- Venezuela
Cleared land for Laotian people 02/23/24
Vannaly is 25 years old. She is a member of the HI Laos demining team and tells us about her experience.
- Laos
With the help of nutrition and communication therapy, Tamara is more independent 02/20/24
Tamara, 5, has cerebral palsy. Thanks to therapy to help her with her nutrition, attention and speech, the little girl is becoming more confident.
- Bolivia and Andean states
Devastating floods hit the Philippines 02/15/24
At least 1.4 million people have been affected and more than 411,000 have been forced to flee due to massive flooding in several regions of Mindanao Island, in the south of the Philippines.
- Philippines
In Ukraine, long-term health consequences of war surface 02/08/24
Two years after the invasion by Russia, Humanity & Inclusion is seeing an increase in the health needs of at-risk populations, particularly those living near the front line in the East of Ukraine.
- Ukraine
Hygiene kits distributed to people with disabilities, acute needs in Ukraine 02/07/24
Caregivers, people with disabilities, and displaced people in Ukraine are receiving hygiene kits from Humanity & Inclusion's teams. These necessities are distributed daily to support those most affected by the war.
- Ukraine
Farming village contaminated by mines in Ukraine 02/06/24
In Ukrainian villages cut off by the war, such as Velyka Komyshuvakha in the Kharkiv region, Humanity & Inclusion is raising awareness of the risks posed by explosive remnants of war.
- Ukraine
After Syria earthquakes, Suzan continues to recover 02/01/24
Suzan’s life was turned upside down by the earthquakes that struck Syria and Turkey in February 2023. Humanity & Inclusion and its partners helped her get through.
- Syria